VisionBlue®: The only trypan blue stain for anterior use approved by the FDA.
No confirmed adverse events reported since launch in 2004.
Highly purified trypan blue with lowest recorded levels of mono-azo dye – known for carcinogenic properties in medical use.
High purification reduces the reported TASS risk from lower purity generic trypan blue use.
Batch level HPLC testing and Certificate of Analysis provides purity assurance and traceability for every lot produced.
FEATURES:
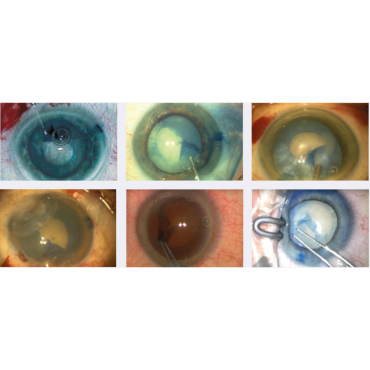
- Assuring the complete, circular capsulorhexis required for a safe phaco-emulsification procedure.
- High success rates in performing anterior capsulotomy in cases of mature or brunescent cataracts.
- Enhanced contrast between the capsulorhexis rim, instrumentation and the adjacent nucleus.
- Reduced risk of radial capsule tears and helps ensure a stable intraocular lens platform.
- Effective and safe recovery of ‘lost’ capsulorhexis edge.
- Improved efficacy in performing capsulorhexis in cases of narrow pupil and pediatric cataracts.
- Improves visualization in cases of vitreous hemorrhage and hazy or scarred cornea.